Drug Cost Comparison Calculator
Compare Brand Name vs Generic Drug Costs
See how much you could save by choosing generic medications. The FDA ensures generics are equally effective as brand-name drugs while costing significantly less.
Savings Potential
Did you know?
Generics cost on average 80-85% less than brand-name drugs. In 2023 alone, generics saved the U.S. healthcare system $313 billion. Most generic drugs cost less than $10 per month.
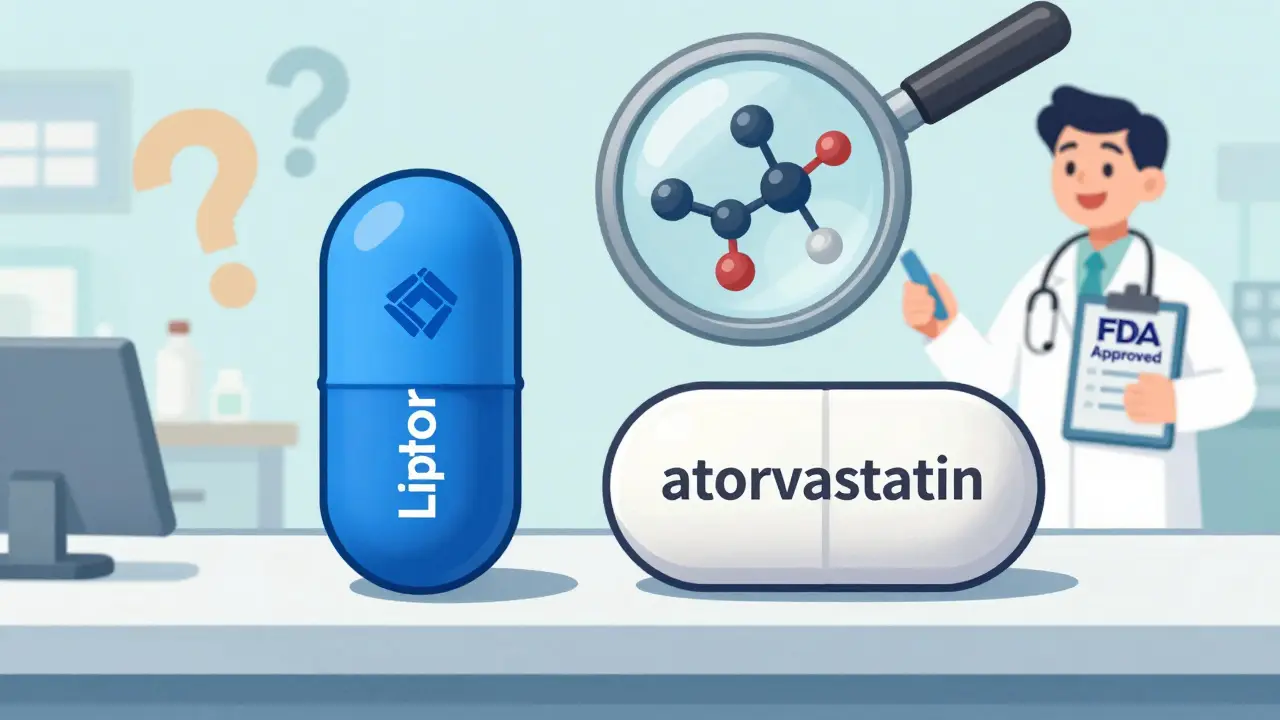
When you pick up a prescription, you might see two different pills on the counter - one with a familiar name like Lipitor, another labeled simply as atorvastatin. You might wonder: is this generic version really the same? Does the label change mean the medicine works differently? The truth is simpler than most people think - and the FDA has spent decades making sure of it.
Same Active Ingredient, Different Look
Generic drugs contain the exact same active ingredient as their brand-name counterparts. That means if you’re taking omeprazole for acid reflux, whether you get Prilosec (the brand) or a generic version, your body gets the same chemical to do the same job. The FDA requires this. No exceptions. The active ingredient must match in strength, dosage form, and how it’s absorbed. That’s not a suggestion - it’s a legal requirement under the Abbreviated New Drug Application (ANDA) process established in 1984.
So why do the pills look different? It’s not about effectiveness - it’s about trademarks. U.S. law says generic manufacturers can’t copy the color, shape, or markings of brand-name drugs. That’s why your generic version might be a white oval instead of a blue capsule. It’s also why the label says omeprazole instead of Prilosec. The brand name is protected; the chemical name isn’t.
Labeling: Exactly the Same, Except the Name
Here’s something most people don’t know: the warning labels, dosage instructions, side effects, and contraindications on generic drug packaging must be identical to the brand-name version. The FDA’s 2021 guidance makes this crystal clear. If the brand-name drug says “May cause dizziness”, so does the generic. If it lists “Avoid with grapefruit juice”, the generic has the same warning.
The only difference? The brand name is replaced with the generic name. No hidden warnings. No cut corners. No extra risks. The FDA reviews every generic label line by line before approval. This isn’t a loophole - it’s a rule.
Therapeutic Equivalence: What the FDA Actually Requires
The biggest myth about generics is that they’re “weaker” or “less reliable.” The science says otherwise. To get approved, a generic drug must prove it’s bioequivalent to the brand. That means in a study with 24-36 healthy volunteers, the generic must deliver the same amount of medicine into the bloodstream - within 80% to 125% - as the brand-name version. That’s not a wide range. It’s tighter than the natural variation between two batches of the same brand-name drug.
Dr. Ameet Nagpal, Director of Pharmacy at Northwestern Medicine, put it plainly: “The 20% variability allowed in bioequivalence studies is actually stricter than what you see within the same brand over time.” In other words, if you took Lipitor last month and again this month, your body might absorb slightly different amounts - and that’s normal. The generic has to match that variability.
Studies back this up. A 2021 analysis in JAMA Internal Medicine looked at over 2 million patients taking generic and brand-name cardiovascular drugs. No difference in heart attacks, strokes, or deaths. A 2023 review in BMJ Open of 47 clinical trials found the same. Generics work the same. Period.

Cost: The Real Difference
The only major difference between generic and brand-name drugs? Price.
Atorvastatin (Lipitor) cost $375.22 per month as a brand in Q2 2023. The generic? $4.00 at Walmart. That’s not a typo. That’s 99% cheaper. For most drugs, generics cost 80-85% less. The Congressional Budget Office estimates generics saved the U.S. healthcare system $313 billion in 2023 alone. Since 2007, that total hits $1.67 trillion.
The FDA says 90% of generics cost less than $10 per month. For people on fixed incomes, on Medicare, or without insurance, that’s not a luxury - it’s the difference between taking medicine and skipping doses. A Kaiser Permanente survey in 2022 found 78% of patients said generic cost savings helped them stick to their treatment plan.
When Generics Need Extra Care
Not all drugs are created equal - in terms of how much room there is for error. For drugs with a narrow therapeutic index (NTI), even tiny changes in blood levels can cause problems. The FDA specifically flags three: warfarin (a blood thinner), levothyroxine (for thyroid disease), and phenytoin (an anti-seizure drug).
For these, switching between brands or between generic manufacturers might require monitoring. A pharmacist at Reddit’s r/pharmacy shared: “I’ve switched thousands of patients to generics with zero issues - except for levothyroxine. We always check TSH levels after the switch.” That’s not because generics are unsafe. It’s because the body is sensitive to small changes in these drugs.
That’s why pharmacists and doctors check the FDA’s Orange Book - a public database that rates drugs for therapeutic equivalence. If a drug is rated “A,” it means it’s interchangeable. If it’s “B,” it’s not. Most generics are rated “A.”

Why Do Some People Say Generics Don’t Work?
You’ll hear stories. “My generic made me dizzy.” “I didn’t feel the same.”
Most of the time, it’s not the drug. It’s the pill.
A University of Michigan study in 2021 found 12% of patients hesitated or worried when they got a pill that looked different - even if it was the same medicine. That anxiety can trigger real symptoms. Placebo effect works both ways. If you believe the generic won’t work, your body might react as if it didn’t.
Drugs.com reviews show generic atorvastatin has a 6.6/10 average rating. Lipitor? 6.3/10. That’s not a gap - it’s a mirror.
What You Can Do
- Ask for the generic. Your doctor can write “Dispense as Written” if they prefer the brand - but unless they do, pharmacists in 49 states can legally switch you to the generic.
- Check the label. Make sure the active ingredient matches what your doctor prescribed. If it does, you’re fine.
- Don’t panic over appearance. A different color or shape doesn’t mean a different drug.
- Monitor for NTI drugs. If you’re on warfarin, levothyroxine, or phenytoin, ask your doctor to check blood levels after switching manufacturers.
The Bottom Line
Generic drugs aren’t second-rate. They’re not cheaper because they’re worse. They’re cheaper because they don’t carry the cost of marketing, patents, or brand-building. The FDA ensures they’re just as safe and effective. In fact, for 90% of prescriptions, they’re the smart choice.
Millions of people use generics every day - and they work. The science is solid. The data is clear. The savings are real. If your doctor prescribes a drug with a generic version, you’re not getting the “discount” version. You’re getting the same medicine - for a fraction of the price.
Are generic drugs as safe as brand-name drugs?
Yes. The FDA requires generic drugs to meet the same strict standards for quality, purity, strength, and safety as brand-name drugs. Every generic must prove it delivers the same active ingredient in the same way as the original. Post-market surveillance through the FDA’s Adverse Event Reporting System (FAERS) shows no higher risk of side effects.
Why do generic drugs look different?
U.S. trademark laws prevent generic manufacturers from copying the color, shape, or markings of brand-name pills. This ensures consumers can tell them apart. These visual differences have zero impact on how the drug works. The active ingredient, dosage, and effectiveness remain identical.
Can I trust a generic drug if it’s made overseas?
Yes. The FDA inspects all manufacturing facilities - whether in the U.S., India, China, or elsewhere - before approving a generic drug. Over 80% of generic drugs sold in the U.S. are made overseas, but they must meet the same quality standards as U.S.-made drugs. The FDA conducts thousands of inspections each year.
Do generic drugs take longer to work?
No. Bioequivalence studies require generics to have the same rate and extent of absorption as the brand-name drug. That means the time it takes to reach peak concentration (Cmax) and total exposure (AUC) must be within 80-125% of the original. If it didn’t, the FDA wouldn’t approve it.
Are there any drugs that don’t have good generic alternatives?
Yes - but they’re rare. Complex drugs like biologics (e.g., insulin, monoclonal antibodies), certain inhalers, and auto-injectors (like EpiPens) are harder to copy exactly. The FDA has approved some biosimilars and complex generics, but they’re not always available. For most common pills - antibiotics, blood pressure meds, statins - generics are plentiful and reliable.
Can I switch between different generic manufacturers?
For most drugs, yes. The FDA considers all generics with an “A” rating in the Orange Book as interchangeable. However, for narrow therapeutic index drugs like levothyroxine or warfarin, your doctor may recommend sticking with one manufacturer to avoid tiny variations in absorption. Always check with your pharmacist if you notice a change in pill appearance.


phyllis bourassa
I swear, my grandma took generics for 20 years and never missed a beat. But then again, she also believed in vinegar for hiccups and cold showers for fever. Still, she lived to 98. Maybe the real miracle isn't the drug-it's that we still trust pills more than our own gut.
Also, why does every pharmacy give me a different color pill? Like, I swear I'm on the same med, but now it's a purple oval instead of a blue round. Feels like my body is being gaslit.